Superior Gut Health Translates to Athletic Power

In this Article
Summary
The key to harnessing an athlete’s power potential is optimizing gut health. By increasing the quality and diversity of gut microbiota, we can increase the power of overall health. This power output propels an athlete to the top of the game via stronger immunity, lower inflammation, enhanced nutrient metabolism, and resilient brain function and behavior.
An athlete’s power traditionally develops from physical training. Power output is critical for success in sport, but power becomes more selective at the highest level, depending on an athlete’s ability and training. If all things are equal physically, what is the key ingredient that will separate athletes at the top of elite sport?
The gut microbiome.
The gut is another route to increase horsepower. Optimizing gut microbiota means increasing the quality and diversity of the microbiota, which can increase the power of overall health. This power output propels an athlete to the top of the game via stronger immunity, lower inflammation, enhanced nutrient metabolism, and resilient brain function and behavior.
While beetroot juice may make the difference between gold and silver, the gut microbiome can determine whether an athlete makes it to the starting line. In this article, I’ll explain how the gut is an athlete’s control center for optimizing their power potential and why this offers the best chance of athletic success.
Why Athletes Need Gut Microbiota
Trillions of microorganisms in the gut powerfully influence our health. They influence nutrient metabolism, immunity, gut development, inflammation, metabolic disease, neurodevelopment, and behavior.1
Gut microbiota’s roles and functions include:
- Enhance digestion and food absorption
- Regulate and strengthen immunity
- Protect from pathogens
- Ferment certain carbohydrates into small chain fatty acids (SCFAs), the energy sources for the liver and muscle cells. They also maintain glycemia during endurance performance, regulate neutrophil function and migration, reduce gut permeability, restrict inflammatory cytokines, and regulate the redox environment.2
- Communicate with the brain.
For an in-depth explanation of how gut microbiota impact our nutrition, read here.
The key to harnessing an athlete’s power potential is optimizing gut health. This means harboring a diverse community of good gut bugs and maintaining this microbial community. Power drops when gut dysbiosis (i.e., microbial imbalance) occurs, resulting from antibiotic use, dietary changes, and most importantly, stress.
In this article, I’ll discuss:
- Stressors on the athlete that hurt the gut
- How gut microbiota alleviate these stressors by strengthening immunity and preventing illness
- A new area of microbiota research involving brain health
Gut Microbiota: A Biotic Shield Against Stressors
Gut health is the athlete’s control center. The following stressors affect optimal gut health:
- Lifestyle (diet)
- Training and competition (intense, prolonged training; psychological)
- Environment (heat, germs, pollutants)
- Travel (jet lag)
- Poor sleep (quantity and quality)
These unavoidable stress factors impact the stability (composition and function) of gut microbiota3 and, consequentially, gut health. This constant attack on the microbiota leads to microbial dysbiosis. For example, exposure to stress can lower gastric emptying and slow transit time in the small intestine.4,5 This reduction in gut motility usually results in bacterial overgrowth.6
Stressors lead to inflammation in the gut, which changes microbial communities for the worse. This creates a cascade effect, hurting nutrient digestion and absorption, immunity, and brain health.
For a further understanding of inflammation and gut health, read here.
The Silent Exercise-Induced Injury That Weakens Immunity
Athletes always end training with an injury. When we think of injuries, we tend to consider the superficial injuries that we see or feel—muscle soreness and bruising, for example.
Deep within the body, however, exercise-induced cellular damage occurs every time an athlete trains or competes hard. Over time, this chronic injury indirectly hurts athletic performance.
Exercise-induced cellular damage is a consequence of different, unavoidable physiological stressors. Examples include:7
- Strenuous exercise
- Heat stress
- Redistribution of blood from circulation through the internal organs to skeletal muscle; blood circulation away from the gut hurts the gut lining and causes gut cell injury
- Oxidative stress and mechanical damage
This injury is quite common. For example, one study found that healthy, young adult male cyclists training 4-10 hours per week experienced a redistribution of blood in just one hour of cycling at 70% of maximum workload capacity.
And that impact was at the healthy human level, not the elite level.
Blood redistribution can cause:
- Lower gastrointestinal (GI) blood circulation
- Increased intestinal permeability where the gut barrier starts to widen and substances from the gut can leak into the bloodstream
- Damage to the small intestine
These inevitable stressors initiate at the protective, one-cell thick barrier of the gut. They cause intestinal cell damage by increasing gut permeability, which opens the door wide open for bad bacteria to enter the bloodstream. If this leaky gut exacerbates, then pro-inflammatory bacterial endotoxemia results in much more serious problems due to immunosuppression.
For example, high amounts of endotoxin can disrupt sleep by activating our body’s defense system. This increases pro-inflammatory markers, cortisol, heart rate, and body temperature. We believe this results from endotoxemia, which releases inflammatory markers. A consistently reported effect of endotoxin on sleep is its suppression of REM sleep, which increases wakefulness and amount of time it takes to go to sleep.
Does this mean the sleep practices at the pro level are fruitless? Not necessarily. It’s just a major factor when considering a solution to the bigger picture. And, once again, it’s evidence as to how maintaining the gut helps an athlete’s power potential: lack of sleep hurts cognition and behavior.
Essentially, exercise-induced stress can weaken the GI barrier and allow bad bacteria to enter the bloodstream, which leads to GI consequences, hydration imbalance, poor absorption of nutrients and electrolytes, and thermal damage to the intestine.
Exercise-induced stress can weaken the GI barrier & allow bad bacteria to enter the bloodstream. Share on XThe result is a subsequent drop in athletic performance.8
The typical nutrition-centered recovery of glycogen repletion, hydration, and muscle repair is not enough to restore performance levels.
The immunological aspect of sports nutrition is often forgotten. It’s just as important to never start a training session with a weak gut as it is to never start a training session while dehydrated.
Immunonutrition via gut microbiota can alleviate the increase in oxidative stress, intestinal permeability, muscle damage, and inflammatory response by strengthening the intestinal barrier. (I’ll discuss this later.) For a further explanation of how the gut strengthens immunity, read here.
Illness Surveillance in Elite Sport Informs Immunonutrition
A weakened immunity opens a wide window of opportunity for illness and a huge cost to training preparation and performance. Common risk factors for athletes include:
- Training loads9,10
- Travel11
- Exercise-induced immune suppression12
- Psychological stress13
- Poor nutrition (energy restriction)14
Collectively, these are stressors. Typically, the respiratory system is the most affected, which occurs in 41-63% of illnesses.15,16 Injury management in the NBA is taking off and so is illness surveillance in elite sport.
A study investigated illness risk factors in athletes nine months before the Rio 2016 Olympic Games. The researchers defined illness as “an event which limited training or competition for greater hours in the prior month.” Here are some of the results:
- Females had the greatest association with illness; the risk was worse when combined with low energy availability.
- Low energy availability, depression symptoms, and high perceived stress were significantly associated with illness.
- Communal living had a three-fold increase in illness rate due to exposure to potential contagious substances.
Because Aquatics has the second largest number of participating athletes at the Olympics, another study investigated the prevalence of illnesses four weeks before, and the incidence of illnesses during, the 2015 FINA World Championships. Results included:
- Of 312 illnesses reported, 17% resulted in time loss.
- Most common illnesses involved the respiratory tract (~34%) or the gastrointestinal tract (~24%) and were caused by infection (~45%).
- During the four weeks before the championships, athletes with illnesses suffered symptoms for eleven days, on average; yet, the average for missing training was only two days.
- About 67% of athletes started the championship with symptoms, and over 50% reported that it affected their performance.
It’s critical to note that athletes continued to train and compete while ill. This has many implications, including increasing the severity of the illness and lowering the quality of performance.
This type of surveillance system—reporting the incidence of illness to identify risk factors—is also gaining momentum in professional tennis. And it’s not just Olympic-level athletes who are exposed to these risk factors; it trickles down to collegiate and elite, recreational exercisers.
The goal of identifying risk factors is to modify risks to prevent the consequential time loss due to illness. Immunonutrition comes into play here.
The imbalance between training and recovery could increase the risk for illness. A systematic review found moderate evidence of a link between training load applied to an athlete and the occurrence of illness. The researchers, however, cautioned that a latent period exists between training load and the onset of an illness. For example, when an athlete experiences a rapid increase in training load, health consequences may not result until 3-4 weeks after the loading.
Illness prevention programs—immunonutrition practices in an athlete’s overall nutritional plan—are critical to optimizing the athlete’s ability to train. And this must be a habitual practice.
Nutrition should go beyond fueling the athlete to enhancing their gut health. Share on XThe goal is to alleviate the severity of the exercise-induced immunodepression phase. This is where nutrition goes beyond fueling the athlete toward designing a gut-enhancing diet.
Think of it this way:
- Reduced Power Output
- Inability to train at high intensity and prolonged duration (loss of quality training)
- Arrival at competition under-prepared (poor health)
- No Power Output
- Loss of training time
- Absence from the starting line
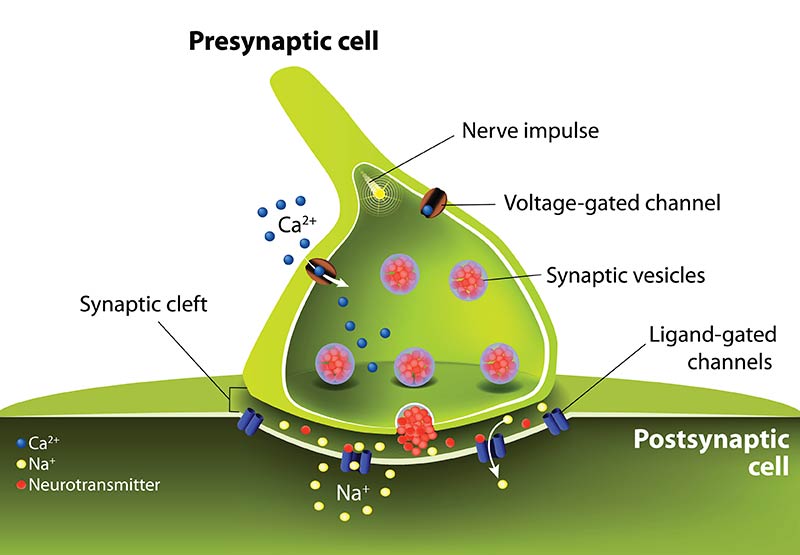
Gut Microbiota: A New Player in Brain Health
Gut microbiota, through crosstalk between the brain and gut microbiome, can influence all parts of physiology, including gut-brain communication, brain function, and behavior.17 We can view gut microbiota as an endocrine organ that has effects outside the gut. This has critical implications for an athlete’s mental health and longevity.
Brain-Gut Axis
The brain-gut axis is a perfect example of bidirectional signaling between two organs—the autonomic nervous system and enteric nervous system (ENS) in the GI tract.
Gut microbiota gain access to the brain through different pathways:
- Neuronal—both the vagus nerve that connects the brain stem to the digestive tract and the ENS
- Endocrine—gut hormones (cortisol) and gut microbiota molecules (SCFAs, tryptophan)
- Immune Signaling—pro-inflammatory cytokines (interleukin-6 (IL-6))17
Gut microbiota can produce neurotransmitters found in the brain, such as serotonin, GABA, noradrenaline, and dopamine.
The brain is also dependent on gut microbes for metabolic products that regulate the brain and behavior. SCFAs are the end products when microbiota ferment complex carbohydrates. Also, cytokines produced in the gut can reach the brain. And the gut microbiota can change the concentration of circulating cytokines, which can impact brain function.18
The hypothalamus-pituitary-adrenal (HPA) is the core regulator for the stress response, which releases stress hormones (norepinephrine, epinephrine) and glucocorticoids (cortisol). Cortisol, the most powerful stress system activator, can not only negatively impact immune cells in the gut by increasing gut permeability and lowering barrier function but also immune cells throughout the body.
Microbiota may control the HPA axis in athletes, and therefore, control hormone release from exercise-induced stress.
This is critical because the HPA axis has a major influence on the brain-gut axis. Psychological or physical stress can dysregulate the HPA axis, which then dysregulates the brain-gut-microbiota axis.
Thus, microbial dysbiosis could have detrimental effects on brain function.
Sleep and Fatigue
The HPA system is critical to balancing the sleep-wake cycle because of its sleep-related hormones.19, 20 Poor sleep negatively affects the HPA axis.21
This leads to an increase in cortisol and changes in the release of testosterone.
For example, the central fatigue hypothesis states that serotonin release is associated with sleep, drowsiness, and central fatigue.22 Low serotonin in the brain can cause mood disturbances and depression,23 and the microbiota impact production24 and regulation25 of serotonin. This latest study discusses how gut microbiota influence serotonin.
Sleep deprivation lowers cognitive function, reaction time, execution, and power potential. Share on XPerformance is typically defined as goal-directed behavior requiring mental effort.21 Cognitive function powers performance, and reaction time and execution are factors of power. Sleep deprivation that lowers cognitive function lowers reaction time and execution and overall power potential.
Psychology
Fatigue and mood disturbances (a critical performance factor) are common among athletes. These include irritability, anxiety, lack of motivation, poor concentration, and depression. The microbiota work by synthesizing and regulating different neurotransmitters and hormones that influence an athlete’s mood, motivation, and feeling of fatigue.26
Microbial manipulation, therefore, may benefit an athlete’s psychology. A study found that supplementing Lactobacillus helviticus and Bifidobacterium longum reduced psychological distress and lowered cortisol levels in humans. It’s speculated that the mechanism behind this alleviates the effects of pro-inflammatory cytokines and oxidative stress.27
Another study found that a multi-species probiotic treatment significantly lowered negative thoughts linked with sad moods. Even though the research is in its infancy, we’re finding that targeting the gut microbiome with probiotic therapy may alleviate or prevent this disrupted brain circuitry.
Neurogenesis
Nutrition’s impact on brain health is gaining speed, especially the focus on new brain cells (neurogenesis), which are critical to the aging athlete. Specifically, polyphenols in the diet can stimulate neurogenesis and improve memory, learning, and cognition.
The cognitive part of sports nutrition must consider exercise-induced psychological stress and its impact on gut microbiota. Normalizing the gut microbiota may help adult neurogenesis28 which is sensitive to stress. This is important for learning and memory. The link between brain plasticity and the gut microbiome is a new avenue of research.
Sports nutrition must consider exercise’s psychological stress and its impact on gut microbiota. Share on XFor example, the gut microbiome is pivotal for the maturation of microglial cells, which are important in neuronal transmission and plasticity (the optimal wiring of neuronal circuits).29 A negative shift in bacterial composition could hurt gut-brain communication, which may lead to a deterioration in neuronal circuits with behavioral consequences.
Essentially, a healthy gut is critical to maintaining good communication along the brain-gut axis that leads to a healthy status. Stress on the central nervous system can affect gut function and lead to microbial dysbiosis.
The brain isn’t just powered by food. A gut-enhancing diet can be a type of nutritional psychiatry that can prevent the dysregulation affecting the brain.
Enhance Your Wattage by Training the Gut
Training the gut is a new sports nutrition paradigm where nutritional strategies, specifically carbohydrates and fluids, are used to induce adaptations in the GI to mitigate GI stress and improve performance. This paradigm shows diet can impact the GI through adaptations.
A nutritional intervention that maintains the gut, however, is another lens to look through when training the gut. This is the power of probiotics, live microorganisms that benefit the host’s health when taken in the correct amount.30
Athletes already have an advantage because those who exercise tend to have a healthier profile of good gut bugs—a diverse microbiota with favorable metabolic and inflammatory profiles.1 Athletes need to maintain this advantage.
To train the gut for a well-functioning GI system, we need to supply our innate microbiome with symbiotic bacteria to harness our gut’s control center. Probiotics are an ideal therapeutic approach to optimize the gut because they interact directly with microbiota.
Of course, the microbe-human relationship is highly complex. There are no established dietary recommendations for probiotic supplementation for athletes,26 and there are only a small number of studies exploring this topic.31 There is modest evidence that probiotics can provide some clinical benefits for athletes.
Probiotics can provide some clinical benefits for athletes. Share on XNot all probiotics are the same—clear communication is pivotal to differentiate products. Key points to consider are:
- Sourcing of recommended products and formulas
- Dose-response requirements for different probiotic strains
- Storage and transport of supplements
- Supplementation timing during travel31
Also, a symbiotic product that combines prebiotics and probiotics is critical because prebiotics feed the probiotics and can enhance the anti-inflammatory benefits.
In my next article, I’ll dive deeper into probiotic supplementation and why we need to move away from the typical “fuel the athlete” mantra that predominantly focuses on macronutrients and calories. The next level of fueling focuses on a gut-enhancing diet. Food impacts our gut microbial composition and function.
Superior gut health is a marginal gain that is pivotal to elite sport. Probiotics are a complementary factor to optimizing the gut microbiome. Harnessing the power of gut health will increase an athlete’s wattage physically and mentally.
References
- Cronin O, O’Sullivan O, Barton W, Cotter PD, Molloy MG, Shanahan F. Gut microbiota: implications for sports and exercise medicine. British Journal of Sports Medicine. 2017; 51(9): 700-701.
- Mach N, Fuster-Botella D. Endurance exercise and gut microbiota: A review. Journal of Sport and Health Science. 2016.
- Galley JD, Bailey MT. Impact of stressor exposure on the interplay between commensal microbiota and host inflammation. Gut Microbes. 2014; 5(3): 390-396.
- Nakade Y, Tsuchida D, Fukuda H, Iwa M, Pappas TN, Takahashi T. Restraint stress delays solid gastric emptying via a central CRF and peripheral sympathetic neuron in rats. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology. 2005; 288: R427-32.
- Wang SX, Wu WC. Effects of psychological stress on small intestinal motility and bacteria and mucosa in mice. World Journal of Gastroenterology. 2005; 11(13): 2016-21.
- Nieuwenhuijs VB, Verheem A, van Duijvenbode-Beumer H, Visser MR, Verhoef J, Gooszen HG, Akkermans LM. The role of interdigestive small bowel motility in the regulation of gut microflora, bacterial overgrowth, and bacterial translocation in rats. Annals of Surgery. 1998; 228(2): 188-93.
- March DS, Marchbank T, Playford RJ, Jones AW, Thatcher R, Davison G. Intestinal fatty acid-binding protein and gut permeability responses to exercise. European Journal of Applied Physiology. 2017; 117(5): 931-941.
- van Wijck K, Lenaerts K, Grootjans J, et al. Physiology and pathophysiology of splanchnic hypoperfusion and intestinal injury during exercise: Strategies for evaluation and prevention. American Journal of Physiology: Gastrointestinal and Liver Physiology. 2012; 303(2): G155–G168.
- Drew MK, Finch C. The Relationship Between Training Load and Injury, Illness and Soreness: A Systematic and Literature Review. Sports Medicine. 2016; 46(6): 861-883.
- Jones CM, Griffiths PC, Mellalieu SD. Training Load and Fatigue Marker Associations with Injury and Illness: A Systematic Review of Longitudinal Studies. Sports Medicine. 2017; 47(5): 943-974.
- Schwellnus MP, Derman WE, Jordaan E, Webb, S. Elite athletes travelling to international destinations >5 time zone differences from their home country have a 2–3-fold increased risk of illness. British Journal of Sports Medicine. 2012; 46(11): 816-821.
- Gleeson M, Williams C. Intense exercise training and immune function. Nestle Nutrition Institute Workshop Series. 2013; 76: 39-50.
- Mann JB, Bryant KR, Johnstone B, Ivey PA, Sayers SP. Effect of Physical and Academic Stress on Illness and Injury in Division 1 College Football Players. Journal of Strength and Conditioning Research. 2016; 30(1): 20-25.
- Gleeson M, Williams C. Intense exercise training and immune function. Nestle Nutrition Institute Workshop Series. 2013; 76: 39-50.
- Engebretsen L, Soligard T, Steffen K, et al. Sports injuries and illnesses during the London Summer Olympic Games 2012. British Journal of Sports Medicine. 2013; 47(7): 407-414.
- Engebretsen L, Steffen K, Alonso JM, et al. Sports injuries and illnesses during the Winter Olympic Games 2010. British Journal of Sports Medicine. 2010; 44(11): 772-780.
- Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nature Reviews Neuroscience. 2012; 13(10): 701-712.
- Dinan TG, Cryan JF. The Microbiome-Gut-Brain Axis in Health and Disease. Gastroenterology Clinics of North America. 2017; 46(1): 77-89.
- Friess E, Wiedemann K, Steiger A, and Holsboer F. The hypothalamic-pituitary-adrenocortical system and sleep in man. Advanced Neuroimmunology. 1995; 5: 111–125.
- Steiger A, Antonijevic IA, Bohlhalter S, Friebos RM, Friess E, and Murck H. Effects of hormones on sleep. Hormone Research. 1998; 49(3-4): 125-130.
- Halson SL. Sleep in elite athletes and nutritional interventions to enhance sleep. Sports Medicine. 2014; 44(1): 13-23.
- Best J, Nijhout HF, Reed M. Serotonin synthesis, release and reuptake in terminals: a mathematical model. Theoretical Biology and Medical Modeling. 2010; 7:34.
- Evans JM, Morris LS, Marchesi JR. The gut microbiome: The role of a virtual organ in the endocrinology of the host. Journal of Endocrinology. 2013; 218(3): 37-47.
- Eisenstein M. Microbiome: Bacterial broadband. Nature. 2016; 533(7603): 104-106.
- Desbonnet L, Garrett L, Clarke G, et al. The probiotic bifidobacteria infantis: An assessment of potential antidepressant properties in the rat. Journal of Psychiatric Research. 2008; 43(2): 164–74.
- Clark A, Mach N. Exercise-induced stress behavior, gut-microbiota-brain axis and diet: a systematic review for athletes. Journal of the International Society of Sports Nutrtion. 2016; 13(1): 43.
- Logan, AC, Katzman, M. Major depressive disorder: probiotics may be an adjuvant therapy. Medical Hypotheses. 2005; 64(3), 533–538.
- Ogbonnaya ES, Clarke G, Shanahan F, Dinan TG, Cryan JF, O’Leary OF. Adult Hippocampal Neurogenesis Is Regulated by the Microbiome. Biological Psychiatry. 2015; 78(4) :e7-9.
- Tognini P. Gut Microbiota: A Potential Regulator of Neurodevelopment. Frontier in Cellular Neuroscience. 2017; 11: 25.
- Health and Nutritional Properties and Guidelines for Evaluation. World Health Organization: Food and Agriculture Organization of the United Nations. 2006.
- Pyne DB, West NP, Cox AJ, Cripps AW. Probiotics supplementation for athletes-clinical and physiological effects. European Journal of Sport Scince. 2015; 15(1): 63-72.


